Covid-19 as a risk factor for the development of stroke
José Enrique Llano-Montalvo, José Francisco Aguilar-Ortiz, Juan Antonio Córdova-Hernández
Unidad Médica Familiar 47, Carlos, Perif. Carlos Pellicer Cámara 8, Casa Blanca 2da Secc, 86060 Villahermosa, Tabasco. Email: j.llano23801@gmail.com.
Abstract
Introduction: The post-acute phase of SARS-CoV-2 infection (long Covid, >90 days) has been associated with vascular sequelae; however, local evidence is required on whether the severity of the acute condition, approximated by hospitalization, increases the subsequent risk of cerebrovascular disease (CVD).Objetive: To determine the association between hospitalization for acute Covid-19 and the subsequent development of stroke.
Materials and methods: A retrospective, analytical, 1:1 matched case-control observational study was conducted in Villahermosa, Tabasco (March 15, 2020–December 31, 2024). Cases were patients with an incident diagnosis of stroke, and controls were patients without stroke, selected from the same population group. Matching was performed by age and sex (caliper ±5 years). Data sources included institutional reporting databases (SINOLAVE) and medical records. Paired odds ratios (ORs) and adjusted ORs were estimated using conditional logistic regression.
Results: One hundred and sixty pairs (n=320) were analyzed. A history of hospitalization for COVID-19 was associated with a higher probability of stroke in the adjusted model (aOR=4.45; 95% CI: 2.40–8.25; p < 0.001). Hypertension showed the strongest association (aOR=6.26; 95% CI: 3.16–12.40; p < 0.001). The main association was consistent in sensitivity analyses for the functional specification of age (OR 4.45–5.24) and stable in LOPO (range 4.25–4.95).
Conclusions: Hospitalization for Covid-19 during the acute phase was independently associated with a higher risk of stroke in the period consistent with prolonged Covid, supporting the need for focused clinical surveillance in patients with severe acute illness.
Keywords: COVID-19, Long COVID, Cerebrovascular Disease, Risk Factors, Case-Control Studies
INTRODUCTION
The SARS-CoV-2 pandemic constituted a global health event of great magnitude, with hundreds of millions of accumulated cases and millions of deaths reported worldwide (WHO, 2023). In Mexico, as of June 25, 2023, 7,633,335 confirmed cases of SARS-CoV-2 infection and 334,336 attributable deaths had been reported, according to national epidemiological surveillance systems. The distribution of the healthcare burden showed a clear predominance of outpatient management (90.43%), while 9.57% of cases required hospitalization, reflecting the proportion of patients who presented severity criteria during the acute phase. In the state of Tabasco, 222,063 confirmed cases and 6,189 deaths were documented, with a hospitalization rate of 6.93%. This epidemiological behavior was observed in a context marked by a high population prevalence of Cardiometabolic comorbidities constitute a set of predisposing factors that increase the risk of clinical complications and worse outcomes associated with SARS-CoV-2 infection (CONAHCYT, CentroGeo, GeoInt, & DataLab, 2023).
At the Mexican Social Security Institute (IMSS), the cumulative care provided during the health emergency reached almost 11 million people, reflecting the healthcare burden and institutional relevance of the problem (IMSS, 2023).
At the same time, cerebrovascular disease (CVD) maintains a substantial burden in terms of mortality and disability. Globally, it is recognized as one of the leading causes of death and disability, with a considerable economic impact (Feigin et al., 2022). In Mexico, cerebrovascular diseases were among the leading causes of death; in 2022, CVD was the seventh leading cause of death nationally, with 35,977 deaths registered (INEGI, 2023). In this scenario, the relevant question is not limited to the acute effects of the infection but extends to its potential persistent vascular consequences in the post-acute phase.
Long COVID (also referred to as post-acute sequelae of SARS-CoV-2 infection) is conceptualized as a chronic systemic state following primary infection, lasting at least three months and with a continuous or fluctuating course (Fineberg et al., 2024). Its clinical importance lies in its heterogeneity, its disabling potential, and its involvement of multiple systems, notably neurological and vascular manifestations (Ong et al., 2023; Rushikesh Ramrao Timewar et al., 2023). The pathophysiological plausibility of cerebrovascular outcomes in the post-acute period is supported by mechanisms of endothelial dysfunction, hemostatic activation, and a prothrombotic/microangiopathic state, with persistent thromboinflammatory phenomena following virological resolution (Ahamed & Laurence, 2022; Giustino et al., 2020; Martins-Gonçalves et al., 2023). Furthermore, it has been proposed that cerebral microvascular alterations and changes in cerebrovascular reactivity could contribute to neurovascular vulnerability in long COVID, with implications for outcomes such as stroke (Fekete et al., 2025; Pommy et al., 2023; Koutsiaris & Karakousis, 2025). Additionally, clinical reviews have highlighted the relationship between COVID-19 and ischemic stroke within the neurological spectrum of the disease (Sagris et al., 2021).
From an epidemiological perspective, there is evidence of an increased risk of stroke following SARS-CoV-2 infection, with findings suggesting gradients according to the severity of the acute illness. In a Danish national cohort, an increased risk of stroke was observed in the acute phase, and this excess risk persisted in the post-infection period among those Cases requiring hospitalization were among those that did not show a long-term increase in stroke rates, while those managed in the community did not (Skov et al., 2024). Nationally, local research on post-COVID stroke has also been documented, although with methodological limitations that do not necessarily resolve the role of the severity of the acute episode (Moreno et al., 2022). Taken together, this background supports the need to specifically estimate whether a history of hospitalization for COVID-19 during the acute phase is associated with a higher occurrence of stroke in the post-acute context, particularly in populations with high exposure and cardiometabolic comorbidities (CONAHCYT, CentroGeo, GeoInt, & DataLab, 2023).
Within this framework, the objective of this study was to determine the association between a history of hospitalization for SARS-CoV-2 infection during the acute phase and the development of cerebrovascular disease in the context of prolonged COVID-19 in patients enrolled at Family Medicine Unit No. 47.
The objective was to determine the association between a history of hospitalization for Covid-19 during the acute phase of the infection and a greater development of stroke, compared to having Covid-19 on an outpatient basis in beneficiaries assigned to UMF No. 47 in the context of prolonged Covid (>90 days).
MATERIALS AND METHODS
The study was conducted in Villahermosa, Tabasco, using data from patients registered at Family Medicine Unit (UMF) No. 47. The design was observational, analytical, matched 1:1 case-control, retrospective, from March 15, 2020, to December 31, 2024. The study population consisted of patients registered at UMF 47 with a history of COVID-19 infection during the period of March 15, 2020, to December 31, 2024. Cases were patients registered at UMF 47 with an incident diagnosis of stroke during the study period. Controls were patients registered at UMF 47 without stroke during the same period, selected from the same population group. A consecutive non-probability sampling was performed using the databases of patients reported in the Online Notification System for Epidemiological Surveillance (SINOLAVE). A minimum sample size was calculated, considering a 1:1 matched study type. A McNemar approach was used, which utilizes an expected proportion of discordant pairs. With these values, the minimum theoretical number of pairs required to detect a difference between discordances was estimated to be estimated with α=0.05 (two-tailed), a power of 80, and assuming an expected OR of 2.05 and a total pair discordance of 0.50. The sample size was calculated using the formula for matched case-control studies with a discordant pairs approach. The matched odds ratio and pair discordance were also calculated. It was established that the minimum size required to guarantee the statistical validity of the study was 133 pairs (266 participants). However, to reduce the impact of potential losses, an additional 10% was added. This adjustment was made to ensure the robustness of the analysis and the representativeness of the sample, increasing the total number of pairs to the required 147 (294 subjects). Inclusion criteria for cases were over 18 years, a history of SARS-CoV-2 infection confirmed by laboratory testing (PCR or rapid test), a first-time diagnosis of stroke, ICD-10 codes (I63, I64, I67, I69), and a diagnosis following a history of SARS-CoV-2 infection with radiological confirmation.
Patients with a history of stroke prior to SARS-CoV-2 infection, stroke diagnosed less than 90 days after SARS-CoV-2 infection, insufficient or incomplete information for follow-up, or referred to another hospital were excluded. This study was conducted at General Hospital No. 46 of the Mexican Social Security Institute in Villahermosa, Tabasco. Information was obtained through the digital health ecosystem from emergency and inpatient medical records. The online notification system for epidemiological surveillance was also consulted to identify all patients registered at the Family Medicine Unit. No. 47 patients who had been notified of COVID-19 (SINOLAVE) at General Hospital No. 46 were included in the study. To identify potential cases, a specific search was conducted for hospital discharged patients with diagnoses compatible with cerebrovascular disease, using ICD-10 coding (Cerebral infarction, acute cerebrovascular accident, and other defined cerebrovascular pathologies). Selected cases were confirmed by reviewing clinical notes and, when available in the medical record, corroborated with imaging studies and/or a clinical description consistent with the diagnosis. To evaluate the development of stroke as a complication following the acute phase of COVID-19, hospitalization during the acute phase was considered the primary exposure. Furthermore, to focus the analysis on the post-acute period compatible with prolonged COVID, the cerebrovascular event was defined as occurring at least 90 days after the COVID-19 episode, as determined by the documented date of diagnosis and/or hospital admission as recorded in the medical record. To ensure temporal comparability and minimize bias arising from differences in observation time, an index date was established for each case-control pair. In cases, this date corresponded to the first confirmed diagnosis of stroke; in controls, the same index date as the matched case was assigned, verifying through documentation that the control remained stroke-free up to that point and had clinical follow-up in the unit. Figure 3 illustrates the case and control selection scheme. Using the obtained sample, a database was created. Subsequently, statistical analysis was performed using R software version 4.5.2. The analysis was conducted in three stages. The first stage involved a systematic evaluation of the database quality, examining the completeness of the information and compliance with the 1:1 matched case-control design. The second stage focused on descriptive and inferential statistics, including confidence intervals and p-values. The bivariate analysis included McNemar's exact test and paired odds ratios (ORs). For the multivariate analysis, conditional logistic regression was used to calculate the adjusted ORs for each variable. The third stage involved complementary evaluation tests to document internal consistency. Standardized differences, sensitivity analysis based on the age variable using 3 models (Linear age, Categorical age, age with natural splines with df=3), the stability of the estimator was also evaluated against possible influential pairs with a LOPO (Leave-one-pair-out) model.
RESULTS
A systematic evaluation of the database quality was conducted to verify internal consistency, completeness of information, and adherence to the 1:1 matched case-control design. All variables included in the analysis had zero missing values. Dichotomous variables were correctly coded in binary format (0 = No, 1 = Yes). The age variable showed plausible value for the analyzed population, maintaining a range consistent with the clinical context and without extreme values suggestive of data entry or measurement errors.
Furthermore, it was confirmed that each pair contained a single case with a confirmed diagnosis of cerebrovascular disease (CVD = 1) and a single control without such a diagnosis (CVD = 0). This preliminary review ensured compliance with the matched design and allowed for bivariate and multivariate analyses conditioned on the matching. Initially, 167 pairs (334 participants) were identified; however, 7 pairs were excluded after applying the exclusion criteria and confirming that they had the necessary information, resulting in a final sample of 160 pairs (320 participants). The characteristics of the study population are described below. The 320 participants comprised 160 pairs with a 1:1 matching ratio, with 160 patients who had experienced a stroke and 160 who had not. The average age was similar in both groups, with a mean of 62.9 years and a standard deviation of ±14.6 years, confirming adequate age balance resulting from the matching. Regarding sex, the distribution was equitable between cases and controls, with 51.9% women and 48.1% men in both groups, indicating complete control of this variable in the study design. The age distribution showed a similar composition in cases and controls, with a predominance of participants ≥65 years old (cases: 44.4%; controls: 45.6%). The median age was comparable (cases: 62.5; controls: 63.0), and the interquartile range was close (cases: 17; controls: 19), reinforcing the age comparability achieved through matching. Regarding the exposures and comorbidities analyzed, a history of hospitalization for COVID-19 during the acute phase was considerably more frequent in the case group (51.2%) compared to the control group (19.4%). Similarly, the prevalence of hypertension (69.4% in cases vs. 29.4% in controls) and diabetes mellitus (44.4% in cases vs. 23.8% in controls) was higher in the case group. In contrast, the proportions of obesity (30.0% vs. 29.4%) were higher in the control group. The prevalence of diabetes (29.4%) and dyslipidemia (28.8% vs. 25.0%) was similar between both groups. Finally, smoking was more frequent in controls (49.4%) than in the cases (37.5%). Table 1.
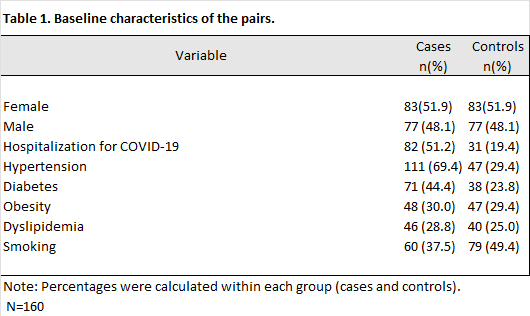
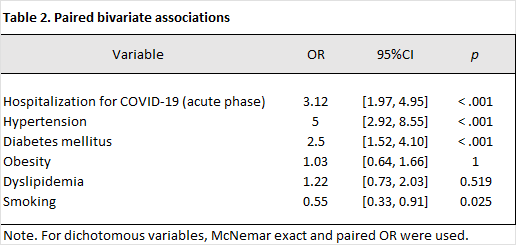
The comparison of the history of hospitalization for Covid-19 and the development of stroke was analyzed using McNemar's test. Bivariate analysis was performed respecting the paired structure for the study type. For age, the paired Wilcoxon test was applied, yielding results without evidence of a statistically significant difference between cases and controls (p = .264). For dichotomous variables, the exact McNemar test was used, and the paired odds ratio (OR) was estimated based on discordant pairs. A positive association was observed between hospitalization for Covid-19 and stroke (OR = 3.12, 95% CI [1.97, 4.95], p < .001), with 99 discordant pairs, providing a large sample size for this estimation. Similarly, hypertension was the variable with the strongest association with the development of stroke, with an odds ratio (OR) of 5.00 (95% CI: 2.92–8.55; p < .001), supported by 96 discordant pairs, along with diabetes mellitus, which also showed a statistically significant association, with an OR of 2.50 (95% CI: 1.52–4.10; p < .001), based on 77 discordant pairs. In contrast, obesity (OR = 1.03, 95% CI [0.64, 1.66], p = 1.000; 67 discordant pairs) and dyslipidemia (OR = 1.22, 95% CI [0.73, 2.03], p = .519; 60 discordant pairs) did not show a statistically significant association. On the other hand, smoking showed an inverse association (OR = 0.55, 95% CI [0.33, 0.91], p = .025; 65 discordant pairs). Furthermore, the effective information for each exposure was corroborated using the number of discordant pairs, as described by their "information ratio" (discordant/pairs). This ratio varied by exposure, being highest for a history of hospitalization (0.62) and hypertension (0.60), thus contextualizing the relative precision of the estimates for each variable. Table 2.
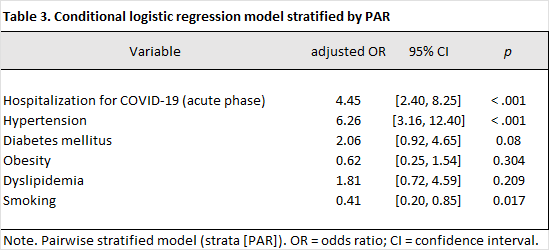
A conditional logistic regression was used, adjusting for major comorbidities, and a history of hospitalization for COVID-19 was found to be associated as an independent risk factor for the development of stroke with an odds ratio (OR) of 4.45 (95% CI: 2.40–8.25; p < 0.001). This result indicates that after controlling confounding variables and covariates, patients with a history of hospitalization for COVID-19 were more than four times more likely to develop stroke. Regarding comorbidities, the most relevant for this study were hypertension, which also demonstrated an independent association with the development of stroke, with an OR of 6.26 (95% CI: 3.16–12.40; p < 0.001), positioning it as one of the most significant risk factors within the analytical model. Diabetes mellitus also showed a moderate positive trend with an OR of 2.06, which, although it did not reach statistical significance (95% CI: 0.92–4.65; p = 0.080), suggests a possible effect that could be confirmed by increasing the sample size. Meanwhile, obesity, with an OR of 0.62; (95% CI: 0.25–1.54, p = 0.3) and dyslipidemia with OR 1.81 (95% CI: 0.72–4.59; p = 0.304) did not show statistically significant associations with the development of stroke in the adjusted model. Relevantly, smoking was associated with a lower risk of stroke (OR 0.41; 95% CI: 0.20–0.85; p = 0.018), a finding that likely reflects residual confounding, selection bias, or survivor bias, rather than a true protective effect. Following the descriptive and inferential analysis, complementary evaluations were conducted to document the model's internal consistency and the robustness of the results. This evaluation was incorporated to demonstrate that the estimated association between a history of hospitalization during the acute phase of a COVID-19 infection and the development of stroke was not due to specific specification assumptions or driven by influential pairs, and that the matching scheme achieved the expected balance in the priori defined variables. Initially, the performance of the matching was evaluated using a descriptive balance diagnosis based on standardized differences (SDN). The results of the post-1:1 matching evaluation showed that the variables used as matching criteria exhibited almost perfect balance. For example, age had an SDN of 0.01 and male sex 0.00, confirming adequate comparability between cases and controls considering these characteristics. In parallel, the behavior of other clinical covariates not used in the matching was described. These showed relevant differences between groups, such as a history of hospitalization for COVID-19 (SMD = 0.67) and hypertension (SMD = 0.80), which is consistent with their differential distribution in a case-control design and corroborates the need to consider these adjustment variables within the multivariate model. Table 3.
In the main model, the OR for hospitalization was 4.45; when categorized by age, the OR was 5.24; and when modeled with splines, the OR was 4.47. Statistical significance was maintained in all three approaches (p < .001), with consistent confidence intervals, which supports the finding that it does not depend on a specific way of incorporating age into the model.
Furthermore, to assess the stability of the estimator against potentially influential pairs, a leave-one-pair-out (LOPO) approach was chosen. This procedure involved re-estimating the conditional model in 160 iterations, excluding a different pair in each iteration, to identify whether the effect size depended disproportionately on specific observations. Importantly, in no iteration did the estimator fall below 1, nor was statistical significance lost (0 iterations with p > .05), confirming the robustness of the estimated effect and ruling out its being conditioned by a limited number of highly influential pairs.
DISCUSSION
The results of this research provide local evidence on the long-term cerebrovascular impact of prolonged COVID-19. The main finding of this study is that a history of hospitalization for COVID-19 acts as a severity marker, quadrupling the probability of developing a stroke (aOR 4.45), independent of age and other classic risk factors for the disease. These data are consistent with the "severity gradient" hypothesis proposed by Skov et al. (2024) and Taquet et al. (2021), who observed that thrombotic risk remains significantly elevated in patients who required hospitalization or intensive care, compared to those managed on an outpatient basis.
When comparing our results with the regional study by Moreno et al. (2022) in Veracruz, our study demonstrated a clear statistical association (p < 0.001). This favorable difference for our research is likely due to the rigor of the matched design and multivariate adjustment, tools that allowed us to eliminate the effect of confounding variables that could have diluted the association in previous, less controlled studies.
A relevant finding is that hypertension (aOR 6.26) was the strongest predictor in our population. This suggests a "double hit" pathophysiological mechanism: an endothelium chronically damaged by hypertension that suffers acute and sustained damage from the systemic inflammation of COVID-19, described by Ahamed and Laurence (2022) as persistent endotheliopathy. In this context, hospitalization for COVID-19 is not only a contributing factor but also an indicator of an inflammatory burden that destabilizes an already vulnerable vascular system.
An important point to highlight is the history of smoking, as it showed an apparent protective effect (aOR 0.41). This finding should not be interpreted as a genuine biological effect, but rather as a manifestation of methodological bias, specifically survival bias. It is plausible that smokers with severe SARS-CoV-2 infection experienced higher mortality during the acute phase, which led to their exclusion from the follow-up cohort for cerebrovascular risk assessment. Consequently, the analyzed population would be predominantly composed of subjects with a lower cumulative smoking burden or with physiological characteristics that confer greater resilience, generating a systematic distortion in the estimate and underscoring the need to interpret this result within a framework of bias control and sensitivity analysis.
Finally, the lack of a significant association with obesity (aOR 0.62) and dyslipidemia (aOR 1.81) in the adjusted model contrasts with classic literature. This could reflect that, in the studied population, there is a limitation derived from the inadequate recording of these conditions in electronic records, propose that the metabolic risk is mainly absorbed by hypertension and diabetes, or that the thrombotic mechanism induced by the virus follows pathways independent of traditional lipid atherosclerosis.
The findings of this study provide evidence that the history of hospitalization for COVID-19 is independently associated with the subsequent development of stroke in the studied population, in the context of prolonged COVID-19, with a more than fourfold increased probability of the event compared to patients who required outpatient management.
A significant interaction with pre-existing comorbidities was established, primarily hypertension, which remains the most relevant baseline risk factor, exacerbating vascular damage in post-COVID patients.
Furthermore, the severity of acute COVID-19 (inferred from the need for hospitalization) has clinical repercussions that extend beyond the acute phase of the disease, corroborating the need to consider stroke as a relevant late complication. For the interpretation of the results, limitations inherent to the retrospective design and the quality of the data sources should be considered. The existence of a potential information bias associated with underreporting in institutional databases is acknowledged, which could lead to an underestimation of comorbidities such as obesity and dyslipidemia. Furthermore, the absence of formal diagnosis or coding during medical care is identified as generating a risk of misclassification, potentially distorting estimates and explaining the lack of statistical significance observed in the multivariate model. Additionally, a relevant limitation was identified in the measurement of the exposure factor: although hospitalization was used as an operational marker of severity, the heterogeneity in the quality of clinical records made it impossible to accurately stratify the level of in-hospital severity, including critical variables such as the need for mechanical ventilation or admission to the ICU. This limitation restricts the possibility of performing a dose-response analysis regarding the impact of clinical severity on vascular risk. Finally, the study is susceptible to survival bias, as its focus was on events occurring in the post-acute stage (>90 days), excluding patients who died during the critical phase of the infection. This is particularly relevant given that it has been established that patients with active COVID-19 generally present with the most severe symptoms and the highest thrombotic risk. Despite the limitations, this study demonstrates methodological rigor that supports the validity of its findings. Key strengths include the matched design and rigorous control of confounding variables. The application of 1:1 matching by sex and age, using caliper matching of ±5 years, ensured optimal comparability between groups, reducing the confounding impact of age, already considered the main risk factor for stroke. Additionally, the analytical rigor distinguishes this study from traditional descriptive approaches. The use of a conditional logistic regression model allowed for the independent estimation of the association between hospitalization for COVID-19 and the development of stroke, while simultaneously adjusting for the effect of clinically relevant covariates such as hypertension and diabetes mellitus. Finally, the rigorous case definition strengthens the study's internal validity. The exclusive inclusion of patients with laboratory-confirmed COVID-19 (PCR or rapid test) and cerebrovascular events verified by imaging and ICD-10 coding minimizes the risk of misclassification, helping to ensure that the results accurately represent the phenomenon analyzed. Given the findings of this study, such as the history of hospitalization as a clinical marker for the development of stroke, the following should be considered: The need for post-acute follow-up in patients with a history of complications during the acute phase of SARS-CoV-2 infection, especially if they have a history of intensive care unit admission. Patient education strategies should be developed for individuals with a history of COVID-19 complications, focusing on the identification of focal neurological symptoms, especially those with comorbidities such as hypertension and diabetes mellitus. It is recommended to strengthen the quality of clinical records to accurately assess risk gradients and thereby improve the precision of future local research.
REFERENCES
1.Ahamed, J., & Laurence, J. (2022). Long COVID endotheliopathy: hypothesized mechanisms and potential therapeutic approaches. The Journal of Clinical Investigation, 132(15). https://doi.org/10.1172/JCI161167
2.CONAHCYT, CentroGeo, GeoInt, & DataLab. (2023). Covid 19- Mexico Board. https://datos.covid-19.conacyt.mx/#DOView
3.Feigin, V. L., Brainin, M., Norrving, B., Martins, S., Sacco, R. L., Hacke, W., Fisher, M., Pandian, J., & Lindsay, P. (2022). World Stroke Organization (WSO): Global Stroke Fact Sheet 2022. International Journal of Stroke, 17(1), 18–29. https://doi.org/10.1177/17474930211065917
4.Fekete, M., Lehoczki, A., Szappanos, Á., Toth, A., Mahdi, M., Sótonyi, P., Benyó, Z., Yabluchanskiy, A., Tarantini, S., & Ungvari, Z. (2025). Cerebromicrovascular mechanisms contributing to long COVID: implications for neurocognitive health. GeroScience, 47, 745–779. https://doi.org/10.1007/s11357-024-01487-4
5.Fineberg, H. V., Brown, L., Worku, T., & Goldowitz, I. (2024). A long COVID definition: A chronic, systemic disease state with profound consequences. National Academies Press. https://doi.org/10.17226/27768
6.Giustino, G., Pinney, S. P., Lala, A., Reddy, V. Y., Johnston-Cox, H. A., Mechanick, J. I., Halperin, J. L., & Fuster, V. (2020). Coronavirus and cardiovascular disease, myocardial injury, and arrhythmia: JACC Focus Seminar. Journal of the American College of Cardiology, 76(17), 2011. https://doi.org/10.1016/J.JACC.2020.08.059
7.IMSS. (2023, May 14). IMSS attended to almost 11 million people during the COVID-19 health emergency declaration. IMSS Social Communication.
8.INEGI. (2023). Statistics of Registered Deaths 2022. https://www.inegi.org.mx/contenidos/saladeprensa/boletines/2023/EDR/EDR2022-Dft.pdf
9.Koutsiaris, A. G., & Karakousis, K. (2025). Long COVID mechanisms, microvascular effects, and evaluation based on incidence. Life, 15. https://doi.org/10.3390/life15060887
10.Martins-Gonçalves, R., Hottz, E. D., & Bozza, P. T. (2023). Acute to post-acute COVID-19 thromboinflammation persistence: Mechanisms and potential consequences. Current Research in Immunology, 4. https://doi.org/10.1016/j.crimmu.2023.100058
11.Moreno, J. E. H., Herrera, J. C., Hernandez, E. L., Parrilla, V. M. P., Guzmán, L. M. R., & Lagunes, Á. A.P. (2022). Cerebrovascular event in post-COVID-19 patients. Ciencia Latina Multidisciplinary Scientific Magazine, 6(5), 4046–4069. https://doi.org/10.37811/CL_RCM.V6I5.3374
12.Ong, I. Z., Kolson, D. L., & Schindler, M. K. (2023). Mechanisms, effects, and management of neurological complications of post-acute sequelae of COVID-19 (NC-PASC). Biomedicines, 11. https://doi.org/10.3390/biomedicines11020377
13.Pommy, J., Cohen, A. D., Patel, M., Umfleet, L. G., Franczak, M., Brett, B. L., Obarski, S., Agarwal, M., Ristow, K., Swanson, S. J., & Wang, Y. (2023). Altered cerebrovascular reactivity along functional networks in older adults with long COVID. Alzheimer's & Dementia, 19. https://doi.org/10.1002/alz.081957
14.Rushikesh Ramrao Timewar, A., Avesh Iliyas Sumar, N., Kohale, N. B., Rathod, S. B., & Manwar, G. G. (2023). Post acute corona virus (Covid-19) syndrome. International Journal of Advanced Research in Science, Communication and Technology, 385–391. https://doi.org/10.48175/ijarsct-8842
15.Sagris, D., Papanikolaou, A., Kvernland, A., Korompoki, E., Frontera, J. A., Troxel, A. B., Gavriatopoulou, M., Milionis, H., Lip, G. Y. H., Michel, P., Yaghi, S., & Ntaios, G. (2021). COVID-19 and ischemic stroke. European Journal of Neurology, 28(11), 3826–3836. https://doi.org/10.1111/ENE.15008
16.Skov, C. S., Pottegård, A., Andersen, J. H., Andersen, G., & Lassen, A. T. (2024a). Short-term and long-term stroke risk following SARS-CoV-2 infection in relation to disease severity: a Danish national cohort study. BMJ Open, 14. https://doi.org/10.1136/bmjopen-2023-083171
17.Skov, C. S., Pottegård, A., Andersen, J. H., Andersen, G., & Lassen, A. T. (2024b). Short-term and long-term stroke risk following SARS-CoV-2 infection in relation to disease severity: a Danish national cohort study. BMJ Open, 14(7), e083171. https://doi.org/10.1136/BMJOPEN-2023-083171
18.WHO. (2023, November 24). COVID-19 Epidemiological Update - 24 November 2023. https://www.who.int/publications/m/item/covid-19-epidemiological-update---24-november-2023
Citation
Llano-Montalvo JE, Aguilar-Ortiz JF, Córdova-Hernández JA, Covid-19 as a risk factor for the development of stroke, ERSJ 2026,1(4) 234-246
Academic Editor: Porfirio Felipe Hernández-Bautista Received: 2026-january-22 Revised: 2026-april-13 Accepted: 2026-april-13 Published: 2026-april-18