Risk factors for latent tuberculosis in contacts of patients with active pulmonary tuberculosis
Rosa del Alba Cardoza-Galmiche, Agustín Flores-Sánchez, Juan Antonio Córdova-Hernández, Dulce Azahar Padilla-Sánchez
Unidad Médica Familiar 47, Perif. Carlos Pellicer Cámara 8, Casa Blanca 2da Secc, 86060 Villahermosa, Tabasco, México. Email: ross.2110@hotmail.com
Abstract
Introduction: Latent tuberculosis Infection (LTBI) is the condition in which a person is infected with Mycobacterium tuberculosis without showing any clinical or radiological signs of active disease. It is estimated that more than a third of the world's population has this infection and that between 5% and 10% will develop the active form, thus contributing to the global burden of the disease.Objetive: To determine the risk factors associated with latent tuberculosis infection (LTBI) in contacts of patients with diagnosis of pulmonary tuberculosis.
Materials and methods: An observational, cross-sectional, and analytical study was conducted between June and December 2024, using the tuberculin skin test (PPD) application census carried out at Family Medicine Unit No. 47. Eighty-two contacts of patients with active pulmonary tuberculosis were included. Data were analyzed using SPSS v26 with descriptive statistics, bivariate analysis, and binary logistic regression.
Results: The prevalence of LTBI was 37.8%, similar to that reported previously. A significant association was observed between having a partner and PPD positivity (p=0.019; OR=1491.78), while male sex showed an inverse association (p=0.011; OR=0.11). Urban origin was associated with a higher probability of infection (p=0.002; OR=8.75).
Conclusions: TB-related illness continues to be a public health problem, highlighting the need to strengthen detection and prevention strategies among contacts of patients with active tuberculosis.
Keywords: Latent tuberculosis Infection; Contacts; Risk factors; contact tracing; public health.
INTRODUCTION
Latent tuberculosis infection is the condition of a person already infected with Mycobacterium tuberculosis (MTb), demonstrated by a positive purified protein derivative for tuberculosis (PPD) test, but without evidence of clinical signs or radiological findings compatible with active disease. It is estimated that more than a third of the population has latent tuberculosis infection (LTBI), and 5% to 10% (Gong, 2021) of the population with latent tuberculosis will develop the disease in its active form. Therefore, reactivation of tuberculosis has represented a serious public health problem in terms of tuberculosis eradication.
Recent data indicate that China has the highest burden of LTBI worldwide, with approximately 350 million people latently infected (Gong, 2021). These figures suggest that a significant percentage of the population with LTBI and the lack of differential diagnosis, along with active tuberculosis (TBa), may be potential reasons for the high morbidity and mortality from tuberculosis in countries with a high burden of this disease. Tuberculosis (TB) is an infectious disease that has caused morbidity and mortality and has affected humanity for over 20,000 years, making it one of the leading causes of death worldwide (Gong, 2021). The disease is caused by a group of bacteria from the order Actinomycetales, family Mycobacteriaceae, specifically the M. tuberculosis (MTb) complex. The bacilli are spread in the environment when an infected person expels them into the air by coughing or speaking. It primarily affects the lungs, but can affect other organs.
Factors such as malnutrition, alcoholism, addictions, impaired immune response, and even poor housing conditions are known to influence the development of the disease.
The objective of this study was to evaluate the risk factors associated with latent TB infection in contacts of patients with active pulmonary tuberculosis.
MATERIALS AND METHODS
The study was conducted at Family Medicine Unit No. 47, IMSS Tabasco, as an analytical cross-sectional observational study from June 1 to December 31, 2024. The study population consisted of 164 contacts of patients with active pulmonary tuberculosis, registered at UMF No. 47. The sample size was calculated in EpiDat using the formula for estimating a proportion in a finite population, considering a 95% confidence level, a 5% margin of error, and an expected proportion based on previous studies (47%), resulting in an estimated total of 115 participants. Contacts of patients with active pulmonary tuberculosis were included, regardless of age or sex, provided they underwent PPD testing. Individuals without contact with patients with active pulmonary tuberculosis and those who did not undergo PPD testing were excluded.
A cross-sectional analytical observational study was conducted using data obtained from a survey administered to residents of Family Medicine Unit No. 47 of the Mexican Social Security Institute (IMSS) in Villahermosa, Tabasco. Data from the PPD vaccination census conducted by the epidemiology department of UMF No. 47 were also reviewed, as well as contact tracing data from the National Epidemiological Surveillance System (SINAVE) platform for identifying risk factors.
From the data obtained, records that did not meet the pre-established inclusion and exclusion criteria due to incomplete information or data inconsistencies were excluded. Following this process, the final sample consisted of 82 individuals who met all the defined methodological criteria and were included in the statistical analysis.
Using the obtained sample, a database was created and analyzed using SPSS version 26, where measures such as prevalence, prevalence ratio, and relative risk were calculated. Additionally, a multivariate binary linear regression analysis was performed to correlate the independent variables with the dependent variable.
RESULTS
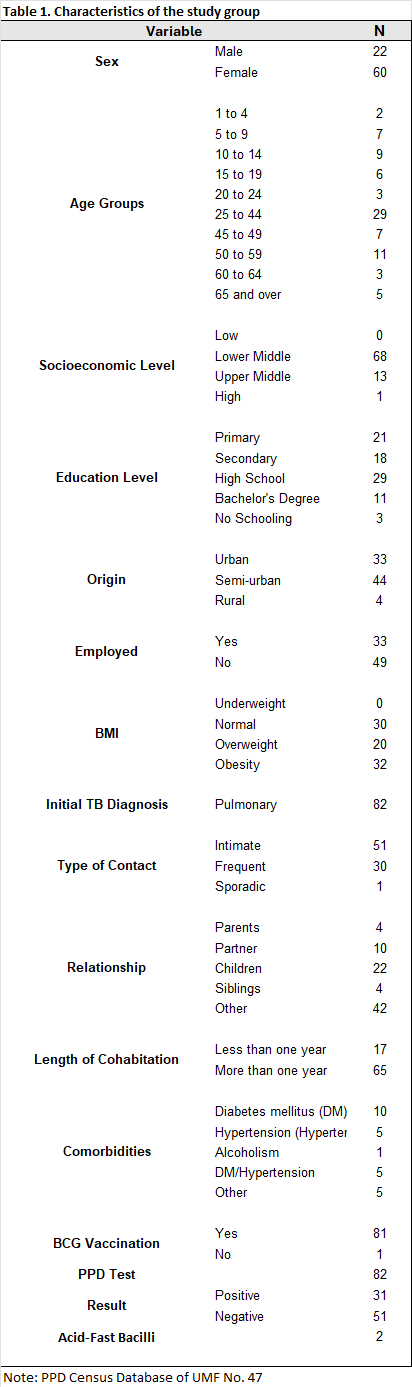
A total of 82 contacts were analyzed. Of these, 26.8% were men and 73.2% were women (Table 1).
The highest level of education was high school for both sexes, while the lowest level of education was for contacts with no schooling.
Regarding the initial diagnosis of the sputum smear-positive patients, 100% had pulmonary involvement. Of the analyzed sample, the variable of the type of contact the participants had with their sputum smear-positive patient showed that intimate contact represented the highest percentage at 62.2%, followed by frequent contact at 36.6%, and lastly, sporadic contact at 1.2%.
According to the length of time the contacts lived with patients with active pulmonary tuberculosis, only 20.7% lived with the patient for less than a year, compared to 79.3% who lived with them for more than a year.
Of the comorbidities identified in the contacts, 39% presented some degree of obesity, followed by 24.4% who were overweight. Among chronic degenerative diseases, the most frequent was diabetes mellitus (DM) at 12.2%, while other comorbidities, such as arthritis or kidney disease, represented 6.1%. Systemic arterial hypertension (SAH) and the coexistence of DM and SAH were also observed in 6.1% of cases. Regarding a history of substance abuse, only one contact (1.2%) reported alcohol consumption (Table 1).
98.9% of the contacts had a history of BCG vaccination. Of the contacts who underwent tuberculin skin testing, 62.2% had a negative result, while 37.8% were positive (Table 2).
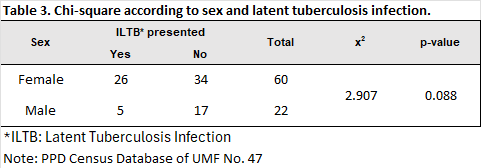
In the bivariate analysis comparing sex and PPD result, a higher proportion of latent tuberculosis infection (LTBI) was observed in women (83.9%) compared to men (16.1%). The chi-square test showed no difference when comparing these two variables (χ² = 2.907; p = 0.888), This indicates that, in the studied population, the risk of developing ILTB is independent of sex. indicating that latent tuberculosis infection does not show a sex predilection (Table 3).
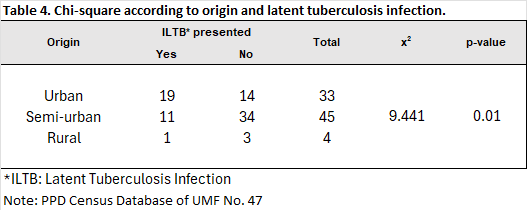
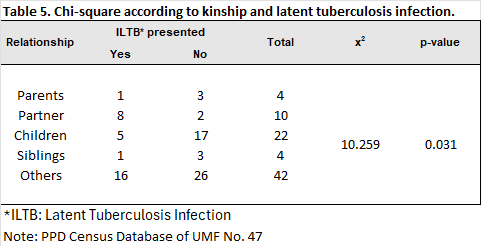
A comparison of latent tuberculosis diagnosis and place of origin revealed a statistically significant difference (χ² = 9.441; p = 0.01). These results suggest that geographic origin significantly influences the occurrence of the event (Table 4). In the group with the kinship variable, the comparison between kinship and the diagnosis of latent tuberculosis infection (LTBI) was statistically significant (χ²=8.624; p=0.031). This indicates that having any type of kinship with a patient with active tuberculosis is a factor for having latent tuberculosis infection (Table 5).
The odds ratio is a measure of association that compares the probability of an event in two different groups, calculated as the ratio of the chances of an event between individuals exposed to a risk factor and those who are not.
The calculation is based on a 2x2 table that classifies individuals as sick/healthy and exposed/not exposed.
For the sex variable, the relative risk estimate was 0.385 (95% CI: 0.125-1.179).
Regarding the origin of the contacts, the risk was 4.185 (95% CI: 1.620-10.809), meaning that those living in urban areas have a four times greater risk of latent tuberculosis infection compared to those living in semi-urban and rural areas.
For the risk assessment related to education level, contacts with only a primary school education had a risk estimate of 1.732 (95% CI: 0.633-4.736), meaning that, compared to other education levels, this population has twice the risk of latent tuberculosis infection.
As for socioeconomic level, those with a middle-to-upper-class level had a risk of 1.509 (95% CI: 0.456-4.988) cruza el 1, and compared to other income levels, they had a 1.5 times greater risk of latent tuberculosis infection.
Regarding the type of contact with sputum smear-positive patients, those with frequent contact had a risk of 1.444 (95% CI: 0.575–3.627) cruza el 1, which, compared to the other types of contact (intimate and sporadic), presented a 1.4 times greater risk of experiencing the studied event.
Of the variable that includes the relationship of contacts to the sputum smear-positive patient, those who are partners of these patients had a risk of 8.522 (95% CI: 1.675–43.353), which showed a significant difference compared to the other types of relationship. This result suggests that the partners of patients with active pulmonary tuberculosis have an 8 times greater risk of developing latent tuberculosis.
Among comorbidities, contacts who were overweight had a risk of 2.567 (95% CI: 0.917–7.185) cruza el 1, meaning that those with this comorbidity have twice the risk of having latent tuberculosis infection (LTBI).
For diabetes mellitus, the risk was 1.769 (95% CI: 0.468–6.687) It shows a trend towards higher risk, however it did not reach statistical significance, which means that those with diabetes mellitus have a 1.7 times greater risk of having LTBI. Contacts with obesity had a risk of 1.510 (95% CI: 0.607–3.755) It shows a trend towards higher risk, however it did not reach statistical significance, meaning they have a 1.5 times greater risk of having LTBI. (Table 6)
The prevalence ratio compares the prevalence of a disease in an exposed group versus an unexposed group and is calculated by dividing the prevalence in the exposed group by the prevalence in the unexposed group.
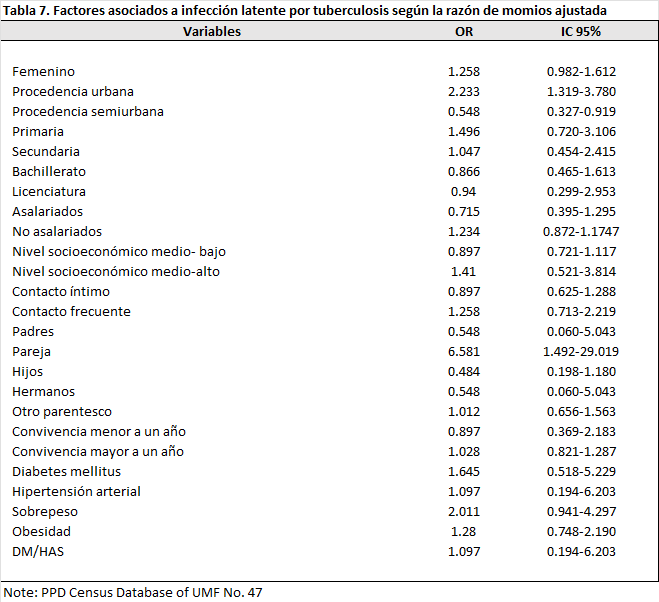
For this analysis, the prevalence ratio between sex and the diagnosis of latent tuberculosis infection (LTBI) was 1.258 (95% CI: 0.982–1.612), suggesting that female participants had a higher prevalence of latent tuberculosis compared to male participants; however, this difference did not reach statistical significance.
According to the origin of the contacts, the prevalence ratio between contacts from urban areas and latent tuberculosis infection was 2.233 (95% CI: 1.319–3.780), and for semi-urban areas it was 0.548 (95% CI: 0.327–0.919), indicating that contacts from urban areas have a higher prevalence of latent tuberculosis compared to those who do not live in such areas. For urban areas, this difference did not reach statistical significance, while for semi-urban areas it did.
Regarding the educational level variable, among contacts with primary education, the prevalence ratio between this group and the PPD result was 1.496 (95% CI: 0.720-3.106); those with secondary education had a prevalence ratio of 1.047 (95% CI: 0.454-2.415); those who completed high school had a prevalence ratio of 0.866 (95% CI: 0.465-1.613); and those with a bachelor's degree had a prevalence ratio of 0.940 (95% CI: 0.299-2.953). Based on these results, the prevalence of TB infection was 1.4 times higher among contacts with only primary education. However, the confidence interval was not statistically significant.
In the group of salaried contacts, the risk was 0.715 (95% CI: 0.395–1.295), while in the group of unsalaried contacts, the risk was 1.234 (95% CI: 0.872–1.1747). Based on these data, the prevalence of LTBI is 1.2 times higher in the unemployed group compared to those with some form of employment, but according to our confidence interval, the data did not reach statistical significance.
The risk was also analyzed according to the socioeconomic level of the contacts. Those in the lower-middle income category had an risk of 0.897 (95% CI: 0.721–1.117), while those in the upper-middle income category had an risk of 1.410 (95% CI: 0.521–3.814). Based on the values obtained, it is interpreted that among contacts with a medium-high socioeconomic level, the prevalence of latent tuberculosis is 1.4 times higher than among those with a medium-low income. However, the confidence interval did not reach statistical significance.
One of the main factors that can contribute to the occurrence of this event is the type of contact with the sputum smear-positive patient. From this analysis, the risk for contacts in the close contact category was 0.897 (95% CI: 0.625-1.288); for those with frequent contact with the patient with active tuberculosis, the risk was 1.258 (95% CI: 0.713-2.219). With this analysis, the prevalence of developing latent tuberculosis infection (LTBI) is 1.2 times higher in those with frequent contact with sputum smear-positive patients than in those with close contact; however, the confidence interval values exceed one, so the data are not statistically significant.
In the kinship category, those who are parents of patients with active tuberculosis had a risk of 0.548 (95% CI: 0.060-5.043); those who are partners, the risk obtained was 6.581 (95% CI: 1.492-29.019); those who are children of sputum-positive patients, the PR was 0.484 (95% CI: 0.198-1.180); as for siblings, the risk was 0.548 (95% CI: 0.060-5.043); and those who fall into the "Other" category, the risk was 1.012 (95% CI: 0.656-1.563). According to our results, it is interpreted that the partners of patients with active tuberculosis have a higher prevalence of latent tuberculosis infection, 2.4 times higher compared to other family members.
Regarding the length of time contacts have lived with sputum smear-positive patients, the prevalence ratio for those who lived with active patients for less than one year was 0.897 (95% CI: 0.369-2.183), while for those who lived with them for more than one year, the prevalence ratio was 1.028 (95% CI: 0.821-1.287). This means that the prevalence of a positive PPD test is 1.028 times higher in the population that has lived with a tuberculosis patient for more than one year than in those who have lived with them for less time. Despite the risk, the confidence interval exceeds one, so it is not statistically significant.
In the section on comorbidities, contacts diagnosed with DM had a prevalence ratio (PR) of 1.645 (95% CI: 0.518-5.229); with HHS it was 1.097 (95% CI: 0.194-6.203); the PR of overweight contacts was 2.011 (95% CI: 0.941-4.297); for obese contacts, the prevalence ratio was 1.280 (95% CI: 0.748-2.190); in contacts with both DM and HHS, a PR of 1.097 (95% CI: 0.194-6.203) was obtained; these data show that those contacts with any comorbidity have a higher prevalence of LTBI than those without. (Table 7)
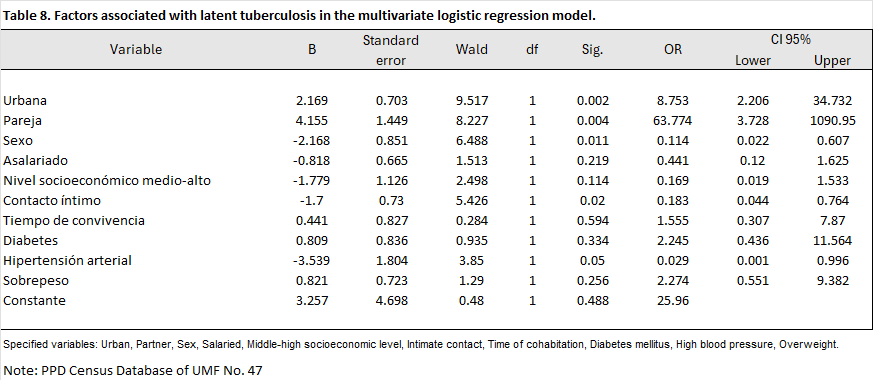
A binary logistic regression analysis was performed to determine the factors associated with the diagnosis of latent tuberculosis infection.
Relevant sociodemographic and clinical variables were included in the model: Male sex showed an inverse association with the occurrence of the event (p = 0.011; OR = 0.11; 95% CI: 0.02–0.61), indicating a lower probability of presentation compared to female sex.
Contacts of urban origin maintained a significant association with a positive result (p = 0.002; OR = 8.753; 95% CI: 2.206–34.732), indicating that people living in urban areas have a significantly higher probability of presenting with latent tuberculosis infection compared to those living in semi-urban and rural areas.
Among those with kinship ties to patients with active tuberculosis, a significant association was observed between those who were partners of these patients (p = 0.004; OR = 63.774; 95% CI: 3.728–1090.950). This suggests that partners of sputum smear-positive patients have a lower risk of developing latent tuberculosis infection (LTBI).
Intimate contact was negatively associated with the occurrence of the event (p = 0.020; OR = 0.18; 95% CI: 0.04–0.76). This finding suggests that maintaining intimate contact significantly decreases the probability of the event occurring. Similarly, systemic arterial hypertension (SAH) was found to be less likely to be associated with the event (p = 0.050; OR = 0.029; 95% CI: 0.001–0.996), decreasing the probability of latent tuberculosis infection.
The variables salaried employment, socioeconomic level, cohabitation time, diabetes mellitus, and overweight did not show statistical significance in this model.
DISCUSSION
In this study, the prevalence of PPD positivity was 37.8% (95% CI: 27.3–48.3), indicating that more than one-third of the participants showed immunological evidence of Mycobacterium tuberculosis infection. This prevalence is consistent with values reported in intermediate-risk populations by various authors. For example, Martínez et al. (2020), in a study of healthcare workers at a public hospital, observed a prevalence of 35.2%, while Hernández et al. (2019) documented 41% in household contacts of patients with TB. Similarly, Gómez et al. (2021) reported prevalences between 30% and 45% in communities with a history of sustained exposure. These figures, together with the present result, reinforce the evidence that latent infection remains a significant problem in exposed populations or those with social conditions that favor transmission. However, the prevalence found was higher than that observed in studies of the general population, such as the one by Sánchez et al. (2018), who reported 18% in individuals with no history of contact. This difference could be explained by the characteristics of the studied population, where factors such as prolonged cohabitation, high housing density, and close contact predominate—elements that increase the probability of exposure to the tuberculosis bacillus. Furthermore, methodological differences—such as the PPD cutoff point, the application technique, or the inclusion of individuals vaccinated with BCG—may also influence the variability between studies. Regarding associated factors, the results showed that having a partner was significantly associated with a higher probability of PPD positivity (p = 0.019; OR = 1491.78; 95% CI: 3.29–674, 638.93). This finding could be related to a greater degree of close contact and prolonged cohabitation, as suggested by Flores et al. (2020), who found that intimate contact and cohabitation were determining factors for the transmission of the infection. Similarly, Castro et al. (2017) identified that the number of cohabitants and the duration of exposure are relevant factors for contagion, especially in family or partner contexts. In contrast, male sex and the presence of hypertension behaved as inversely associated factors, although the latter could be influenced by age or a confounding effect, as also discussed by Mendoza et al. (2020) in studies of comorbidities and immunological susceptibility. Regarding sex, several authors have reported differences in the immunological response to the PPD test, observing greater reactivity in women, possibly due to a hormonal effect or greater access to health services, which increases detection. This pattern agrees with the present study, where male sex was associated with a lower probability of positivity, a result that coincides with that described by Ramos et al. (2022) and Hernández et al. (2019). On the other hand, although the multivariate model included variables such as educational level, occupation, and nutritional status, none showed a significant association. This behavior has also been reported in similar studies, where structural determinants (such as socioeconomic level) influence PPD positivity indirectly, through housing conditions and access to health services, rather than through a direct effect. Taken together, the findings confirm that LTBI remains a relevant health problem, even in populations without active disease, and that interpersonal exposure factors play a key role in its persistence. The differences found with other studies can be attributed to sample composition, population size, heterogeneity in diagnostic criteria, and local sociodemographic characteristics. Finally, it is important to acknowledge that the limited sample size (n=82) and the presence of wide confidence intervals for some variables may have affected the precision of the estimates, which constitutes a methodological limitation. However, the overall consistency with previous studies supports the validity of the results and underscores the need to strengthen detection and prevention strategies for latent tuberculosis infection in community and family settings. This research allowed us to determine the prevalence of latent tuberculosis infection (LTBI) using the PPD test and to analyze associated factors in a specific population. The results obtained reflect that LTBI continues to be a current public health problem, especially in groups with prolonged exposure or in contexts where living conditions facilitate the transmission of Mycobacterium tuberculosis. The PPD positivity rate was 37.8% (95% CI: 27.3%–48.3%), meaning that more than one-third of the individuals tested had latent tuberculosis infection. This value is higher than that reported in general population studies, but similar to that found in research conducted in intermediate-risk populations or those with a history of contact with active tuberculosis cases. This suggests that continuous exposure, close cohabitation, and socio-environmental conditions can significantly influence the probability of infection, even in the absence of active disease. In the multivariate analysis, three factors of interest were identified: having a partner, male sex, and the presence of hypertension. The variable "having a partner" showed a positive and significant association with PPD positivity, which could be explained by a higher frequency of close contact, prolonged cohabitation, or indirect exposure to risk environments through the partner or family. This result reinforces the importance of the home environment as a transmission site and the need to strengthen screening and follow-up of intrafamilial contacts. On the other hand, male sex and hypertension behaved as inversely associated factors. Although the relationship between sex and susceptibility to tuberculosis infection is not entirely consistent across studies, some authors have suggested that immunological and hormonal differences may influence the response to the PPD test, in addition to possible biases in exposure or detection. In the case of hypertension, its apparent protective effect could be explained by lower exposure or by confounding factors related to age or more frequent medical care; therefore, this result should be interpreted with caution and explored in subsequent studies. In contrast, variables such as educational level, occupation, body mass index, diabetes mellitus, and alcohol consumption did not show a significant association with the PPD result. However, it is possible that these factors act indirectly, through structural conditions or social determinants that were not fully captured in this analysis. The study had limitations inherent to the sample size (n = 82), which may have affected the stability of some estimates and increased the width of the confidence intervals. Nevertheless, the model showed an adequate fit and allowed for the identification of epidemiologically relevant associations. Furthermore, the cross-sectional nature of the study prevents the establishment of causal relationships; therefore, longitudinal research is recommended to confirm the direction of the associations found. Despite these limitations, the results contribute to understanding the magnitude and determinants of latent tuberculosis infection in local populations. The findings highlight the need to strengthen early detection, surveillance, and prevention strategies, especially in groups with risk factors for continuous exposure or close contact. They also underscore the importance of incorporating latent tuberculosis infection screening into tuberculosis control programs, not only for index cases but also for their contacts and the community at large. Finally, this study reaffirms the relevance of addressing tuberculosis from a comprehensive perspective, considering the social and familial determinants that influence the transmission and persistence of the infection. The implementation of preventive actions, health education, and sustained epidemiological surveillance are key elements for reducing the burden of latent infection and preventing progression to active forms of the disease, thus contributing to the achievement of national and international tuberculosis control goals. Based on the results obtained in this research on latent tuberculosis infection (LTBI) and their comparison with available scientific evidence, the following recommendations are made to strengthen tuberculosis prevention, detection, and control measures at the local and community levels. These suggestions aim to translate the study's findings into practical strategies that contribute to reducing the transmission of Mycobacterium tuberculosis and the progression of latent infection to active disease: 1. Strengthen screening strategies for LTBI. It is necessary to implement ongoing LTBI detection campaigns using the PPD test in high-risk groups, especially household members and partners of patients with active tuberculosis. This would allow for the timely identification of infected individuals and reduce progression to active disease. 2. Promote medical follow-up and epidemiological surveillance of contacts. The study results show that close cohabitation is a determining factor for PPD positivity. Therefore, it is recommended to establish protocols for tracing and monitoring household contacts, prioritizing those with frequent or prolonged exposure. 3. Strengthen health education and health communication. It is essential to develop educational interventions aimed at the general population and at-risk groups that promote understanding of the transmission, prevention, and treatment of latent tuberculosis infection. Health education can help reduce stigma and increase adherence to screening and control programs. 4. Train health personnel in the identification and management of latent tuberculosis infection (LTBI). It is recommended to strengthen the ongoing training of medical and nursing staff in the reading, interpretation, and follow-up of the PPD test, as well as in the appropriate management of positive cases and their timely referral to specialized care units. 5. Incorporate LTBI detection into institutional and community programs. Given that latent infection represents a significant reservoir of transmission, its inclusion in tuberculosis control programs is a priority. Integrating screening into community campaigns or occupational health assessments could expand coverage and reduce the risk of outbreaks. 6. Promote local investigation and continuous surveillance. Studies with larger samples and longitudinal designs are recommended to establish causal relationships and evaluate the impact of identified associated factors. Active surveillance is also necessary to understand local trends in the prevalence and resistance of Mycobacterium tuberculosis. 7. Consider the social determinants of health in control strategies. Given that transmission is related to housing conditions, overcrowding, and access to health services, it is suggested that an intersectoral approach be incorporated that includes improving social and environmental conditions as an integral part of the fight against tuberculosis.
REFERENCES
1. Basto-Abreu, A., López-Olmedo, N., Rojas-Martínez, R., Aguilar-Salinas, C. A., Moreno-Banda, G. L., Carnalla, M., Rivera, J. A., Romero-Martínez, M., Barquera, S., & Barrientos-Gutiérrez, T. (2023). Prevalence of prediabetes and diabetes in Mexico: Ensanut 2022. Salud Pública de México, 65, s163–s168. https://doi.org/10.21149/14832 2. Campbell, J. R., Winters, N., & Menzies, D. (2020). Absolute risk of tuberculosis among untreated populations with a positive tuberculin skin test or interferon-gamma release assay result: Systematic review and meta-analysis. The BMJ, 368. https://doi.org/10.1136/bmj.m549CTO. (2011). CTO Manual, Infectology. (8th ed.). CTO Editorial Group. 3. Fabela Rodríguez, J. (2008). Prevalence of Latent Tuberculosis Infection in Contacts of Patients with Active Pulmonary Tuberculosis. Autonomous University of Nuevo León. 4. Gong, W., & Wu, X. (2021). Differential Diagnosis of Latent Tuberculosis Infection and Active Tuberculosis: A Key to a Successful Tuberculosis Control Strategy. In Frontiers in Microbiology (Vol. 12). Frontiers Media S.A. https://doi.org/10.3389/fmicb.2021.745592 5. Latent Tuberculosis Infection. Updated and unified guidelines for programmatic management. (2018). 6. Global Tuberculosis Report 2021. (n.d.). http://apps.who.int/bookorders. 7. National Institute of Public Health (Mexico), J. A., National Center for Health Information and Documentation (Mexico), M., Flores-Valdez, M. A., & Hernández-Pando, R. (2010). Public health in Mexico. In Public Health of Mexico (Vol. 52, Number 1). National Institute of Public Health. http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S0036-36342010000100011&lng=es&nrm=iso&tlng=es 8. Kaul, S., Nair, V., Birla, S., Dhawan, S., Rathore, S., Khanna, V., Lohiya, S., Ali, S., Mannan, S., Rade, K., Malhotra, P., Gupta, D., Khanna, A., & Mohmmed, A. (2022). Latent Tuberculosis Infection Diagnosis among Household Contacts in a High Tuberculosis-Burden Area: a Comparison between Transcript Signature and Interferon Gamma Release Assay. Microbiology Spectrum, 10(2). https://doi.org/10.1128/spectrum.02445-21 9. Mycobacteriosis: Manual of Standardized Procedures for Epidemiological Surveillance of Tuberculosis and Leprosy. (n.d.). 10. Pando, R. H., & Barrios-Payán, J. (2013). Latent Tuberculosis (Vol. 56). 11. Paz-Ayar Nibardo, Mejía-Rodríguez Ivonne, García-Velasco Leopoldo, Alcalá-Martínez Enrique, Martínez-Vivar Juan Carlos, & Niebla-Fuentes María del Rosario. (2018). Economic Determinants of Tuberculosis Incidence in Mexico. Revista de sanidad militar, 72, 5–6. 12. PC, & SPPS. (n.d.). SECRETARY OF HEALTH Mexican Official STANDARD NOM-006-SSA2-2013, For the prevention and control of tuberculosis. 13. Reichler, M. R., Khan, A., Yuan, Y., Chen, B., McAuley, J., Mangura, B., & Sterling, T. R. (2020). Duration of exposure among close contacts of patients with infectious tuberculosis and risk of latent tuberculosis infection. Clinical Infectious Diseases, 71(7), 1627–1634. https://doi.org/10.1093/cid/ciz1044 14. Reichler, M. R., Khan, A., Yuan, Y., Chen, B., McAuley, J., Mangura, B., Sterling, T. R., Bakhtawar, I., LeDoux, C., McAuley, J., Beison, J., Fitzgerald, M., Naus, M., Nakajima, M., Schluger, N., Hirsch-Moverman, Y., Moran, J., Blumberg, H., Tapia, J., … Luo, C. (2020). Duration of Exposure Among Close Contacts of Patients with Infectious Tuberculosis and Risk of Latent Tuberculosis Infection. Clinical Infectious Diseases, 71(7), 1627–1634. https://doi.org/10.1093/cid/ciz1044 15. Sagili, K. D., Muniyandi, M., Shringarpure, K., Singh, K., Kirubakaran, R., Rao, R., Tonsing, J., Sachdeva, K. S., & Tharyan, P. (2022). Strategies to detect and manage latent tuberculosis infection among household contacts of pulmonary TB patients in high TB burden countries - a systematic review and meta-analysis. Tropical Medicine & International Health, 27(10), 842–863. https://doi.org/10.1111/tmi.13808 16. Vargas, R., Ríos, N., & Salazar, L. (2003). Tuberculosis care costs: The case of the National Institute of Respiratory Diseases (INER). Rev Inst Nal Enf Resp Mex, 16(4), 219–225. 17. Velen, K., Shingde, R. V., Ho, J., & Fox, G. J. (2021). The effectiveness of contact investigation among contacts of tuberculosis patients: a systematic review and meta-analysis. European Respiratory Journal, 58(6), 2100-266. https://doi.org/10.1183/13993003.00266-2021.
Citation
Cardoza-Galmiche RA, Flores-Sánchez A, Córdova-Hernández JA, Padilla-Sánchez DA, Risk factors for latent tuberculosis in contacts of patients with active pulmonary tuberculosis , ERSJ 2026,1(4) 218-233
Academic Editor: Gabriel Valle- Alvarado Received: 2026-January-12 Revised: 2026-april-09 Accepted: 2026-april-09 Published: 2026-april-18